What is the Allergy Rhinitis Drugs Market Size in 2026?
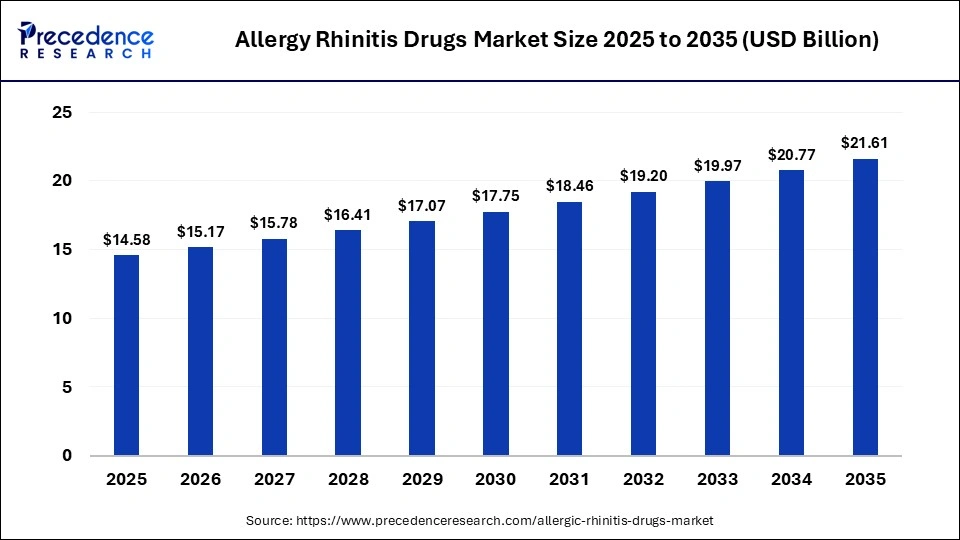
The global allergy rhinitis drugs market size was calculated at USD 14.58 billion in 2025 and is predicted to increase from USD 15.17 billion in 2026 to approximately USD 21.61 billion by 2035, expanding at a CAGR of 4.01% from 2026 to 2035. The list of drugs that treat allergic rhinitis is steadily rising, fueled as much by heightened exposure to pollen, urban air pollution, and new-to-the-public awareness of respiratory allergies. This trend is driving demand for antihistamines, corticosteroid nasal sprays, and immunotherapy as branded or generic alternatives.
Key Takeaways
- North America held a major market share of 37% in 2025.
- Asia-Pacific is expected to host the fastest-growing market in the coming years.
- By disease type, the perennial allergic rhinitis segment contributed the biggest revenue share of the market in 2025.
- By disease type, the seasonal allergic rhinitis segment is expected to expand rapidly in the market in the coming years.
- By treatment type, the antihistamines segment registered its dominance over the global market in 2025.
- By treatment type, the immunotherapy segment is expected to witness the fastest growth in the market over the forecast period.
- By drug type, the prescription segment led the market in 2025.
- By drug type, the OTC segment is expected to grow with the highest CAGR in the market during the studied years.
- By route of administration, the oral segment contributed the biggest revenue share of the market in 2025.
- By route of administration, the intravenous segment is expected to be the fastest-growing segment in the market.
