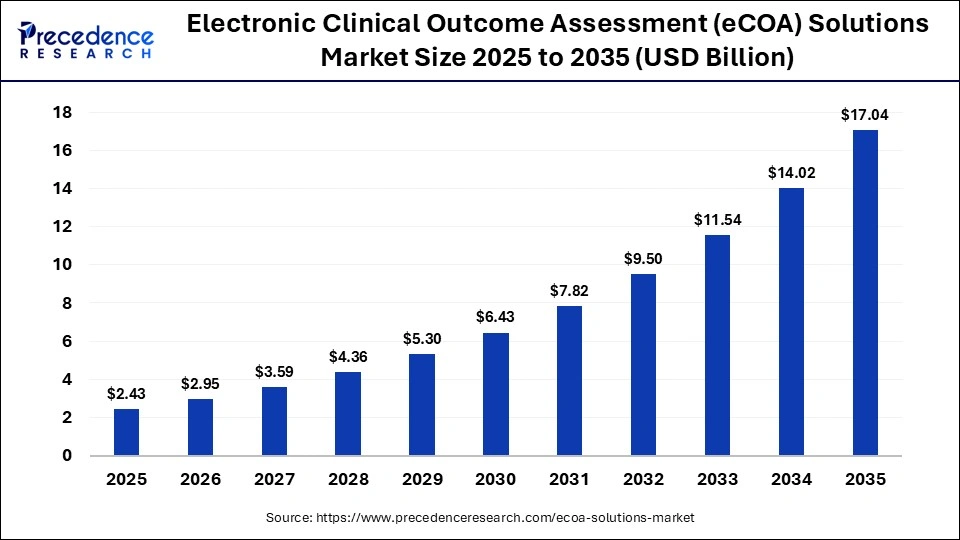
The global electronic clinical outcome assessment (eCOA) solutions market size to expand from USD 2.43 billion in 2025 to USD 17.04 billion by 2035. The market will grow at a compound annual growth rate (CAGR) of 21.50% from 2026 to 2035, propelled by surging adoption of decentralized clinical trials, regulatory support for digital endpoints, and demand for real-time patient data capture.
 Get this report to explore global market size, share, CAGR, and trends, featuring detailed segmental analysis and an insightful competitive landscape overview @ https://www.precedenceresearch.com/sample/7313
Get this report to explore global market size, share, CAGR, and trends, featuring detailed segmental analysis and an insightful competitive landscape overview @ https://www.precedenceresearch.com/sample/7313
Electronic Clinical Outcome Assessment (eCOA) Solutions Market Key Insights
- North America holds 37% market share in 2025, leading due to high R&D spending and mature digital infrastructure.
- Software platforms dominate with 55% share, while Asia Pacific emerges fastest at 21.50% CAGR amid rising trial outsourcing.
- Oncology claims 28% revenue in therapeutic areas, with pharmaceutical & biotech firms capturing 45% end-user share.
- Cloud/SaaS leads deployment at 60%, and mobile apps top devices at 50%.
Market Revenue Overview
| Metric | Value (USD Billion) | Notes |
|---|---|---|
| 2025 Market Size | 2.43 | Base year accounted |
| 2026 Projected | 2.95 | Start of forecast period |
| 2035 Projection | 17.04 | End of forecast at 21.50% CAGR |
| North America 2025 | 0.90 | 37% global share |
| Asia Pacific 2035 | 4.17 | Fastest growth region |
AI’s Role in Electronic Clinical Outcome Assessment (eCOA) Solutions
Artificial intelligence enhances eCOA by analyzing vast datasets in real-time to spot patient response trends and customize workflows via machine learning. Predictive models reduce dropouts, while natural language processing handles unstructured feedback for better data quality and trial efficiency.
Electronic Clinical Outcome Assessment (eCOA) Solutions Market Key Growth Drivers
Integration of wearables enables continuous monitoring, boosting adoption in patient-centric trials. Cloud platforms offer scalability and security, while user-friendly mobile tools improve engagement and reduce errors. Regulatory alignment from FDA and EMA, plus AI analytics, accelerates standardized digital endpoints across global studies.
Why Do Software Platforms Dominate eCOA?
Software platforms captured 55% share in 2025 by providing centralized, cloud-based capture of patient-reported, clinician-reported, and observer-reported outcomes. They ensure real-time visibility and compliance in decentralized trials.
Why Are Cloud/SaaS Solutions Leading Deployment?
Cloud/SaaS held 60% share in 2025 for enabling global consistency, real-time data access, and automated reminders that cut protocol deviations. Hybrid models grow at 21% CAGR to balance accessibility with local compliance needs.
Electronic Clinical Outcome Assessment (eCOA) Solutions Market Regional Analysis
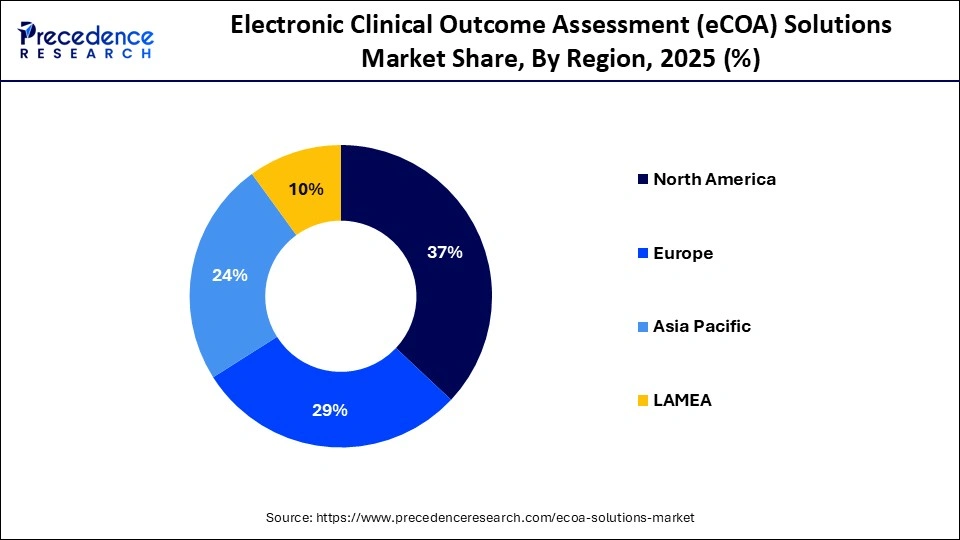
North America leads with USD 899.10 million in 2025, reaching USD 6.39 billion by 2035 at 21.67% CAGR, fueled by U.S. innovation in oncology and rare diseases.
Asia Pacific surges from USD 583.20 million to USD 4.17 billion, driven by China’s infrastructure and India’s outsourcing.
Europe grows from USD 704.70 million to USD 5.03 billion at 21.71% CAGR, with Germany excelling in GDPR-compliant deployments.
Segmentation Highlights
-
Component: Services grow fastest at 22% CAGR for implementation and support in hybrid trials.
-
Device: Wearables hit 23.70% CAGR, complementing mobile apps for objective data.
-
Therapeutic Area: Rare diseases expand at 24% CAGR for dispersed patient tracking.
-
End-User: CROs rise at 23.40% CAGR amid outsourcing in decentralized studies.
Electronic Clinical Outcome Assessment (eCOA) Solutions Market Top Companies
Key players include
- AssisTek
- Climedo Health GmbH
- Clinical Ink
- CRF Health
- EvidentIQ
- ICON plc
- IQVIA
- Integra IT S.A.S
- Kayentis.
Recent Breakthroughs
-
YPrime launched Advanced eCOA Oversight in October 2025 for PI data review per EMA guidelines.
-
SuSuvoda’s IRT earned Leader status in December 2025 for trial supply management.
-
Medable’s Innovation Workshop in December 2025 advanced AI trial tech with FDA input.
You can place an order or ask any questions. Please feel free to contact us at sales@precedenceresearch.com |+1 804 441 9344