What is the CAR-T Therapies for Blood Cancers Market Size in 2026?
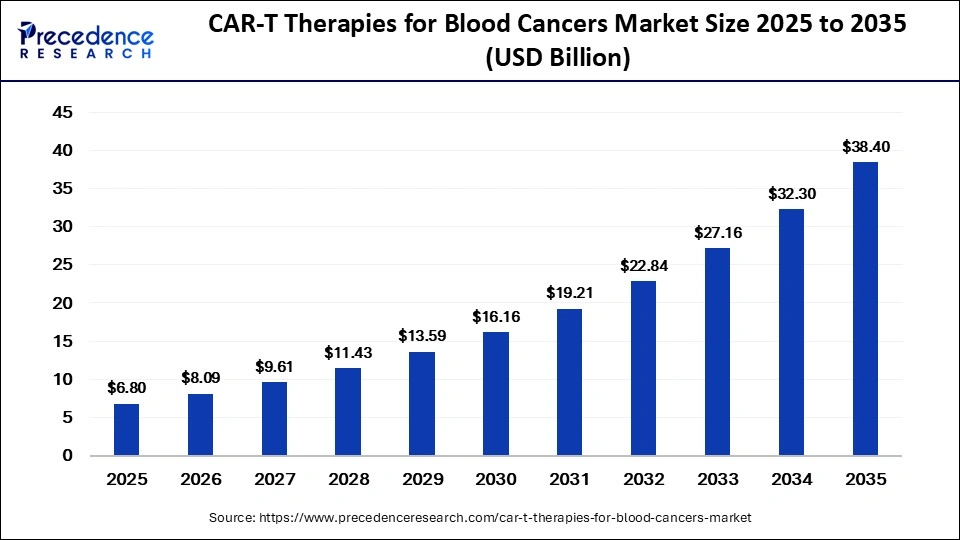
The global CAR-T therapies for blood cancers market size was valued at USD 6.80 billion in 2025 and is projected to grow from USD 8.09 billion in 2026 to approximately USD 38.40 billion by 2035, registering a CAGR of 18.90% during the forecast period from 2026 to 2035. The market growth is primarily driven by the increasing success of CAR-T cell therapies in treating resistant and relapsed blood cancers, along with the rising demand for personalized and targeted treatment approaches.
Key Takeaways
- North America dominated the CAR-T therapies for blood cancers market with a share of 50% in 2025.
- Asia Pacific is expected to grow at the fastest CAGR of 22.5% during the forecast period.
- By target antigen, the CD19-targeted CAR-T segment held a 45% share of the market in 2025.
- By target antigen, the BCMA-targeted CAR-T segment is the second-largest shareholder and is expected to grow at a 21.5% CAGR between 2026 and 2035.
- By cancer type, the diffuse large B-cell lymphoma (DLBCL) segment accounted for a major share of 35% in the market in 2025.
- By cancer type, the multiple myeloma segment is expected to grow at the fastest CAGR of 22% between 2026 and 2035.
- By therapy type, the autologous CAR-T segment led the global market with a share of 80% in 2025.
- By therapy type, the allogeneic CAR-T segment is the second-largest shareholder and is expected to grow at a 25% CAGR between 2026 and 2035.
- By end-use, the hospitals segment held a 50% market share in 2025.
- By end-use, the specialty cancer treatment centers segment is the second-largest shareholder and is expected to grow at a 20.5% CAGR between 2026 and 2035.
- By manufacturing type, the in-house manufacturing segment led the global market with a share of 60% in 2025.
- By manufacturing type, the contract manufacturing segment is expected to grow at a 21.5% CAGR between 2026 and 2035.
Market Overview
The CAR-T therapies for blood cancers market focuses on the development and commercialization of advanced immunotherapies designed to treat blood-related cancers such as leukemia, lymphoma, and multiple myeloma. CAR-T therapy works by modifying a patient’s own T-cells to recognize and destroy cancer cells more effectively, making it a highly targeted and personalized treatment approach.
The rising global prevalence of blood cancers is a major factor supporting market growth. CAR-T therapies are gaining significant attention due to their precise mechanism of action and strong clinical outcomes in patients with relapsed or treatment-resistant cancers. In addition, increasing investments by pharmaceutical and biotechnology companies in research and development of innovative cell therapies are expected to further accelerate market expansion in the coming years.
Impact of AI on the CAR-T Therapies for Blood Cancers Market
Artificial intelligence (AI) is playing a significant role in advancing the CAR-T therapies for blood cancers market by improving the speed, accuracy, and efficiency of therapy development. AI-powered technologies can rapidly analyze large genetic and clinical datasets to identify tumor-specific antigens, enabling more precise target discovery and optimized CAR-T cell design.
In addition, machine learning algorithms help identify patients who are most likely to respond positively to CAR-T therapy, supporting personalized treatment strategies. AI-driven tools also enhance T-cell engineering, optimize gene-editing techniques, and streamline manufacturing processes, leading to improved production speed, consistency, and product quality. These advancements are helping accelerate innovation and expand the clinical potential of CAR-T therapies for blood cancers.
CAR-T Therapies for Blood Cancers Market Trends
- With the growing burden of hematologic malignancies, healthcare providers are increasingly using CAR-T cell therapies for their targeted and personalized approaches.
- Increasing FDA approvals for expanded indications and earlier-line use of CAR-T therapies are improving patient access and accelerating global market adoption.
- Advancements in gene-editing technologies like CRISPR-Cas are enabling the development of safer, more efficient, and effective CAR-T therapies, expanding their therapeutic potential.
- Growing collaborations between biotech and pharmaceutical companies, along with rising R&D investments, accelerating product development, scaling manufacturing, and strengthening commercialization to meet rising demand.
Market Dynamics
Drivers
Demand for Personalized Treatment
The increasing demand for personalized therapies is a key factor driving the growth of the CAR-T therapies for blood cancers market. CAR-T cell therapy is transforming blood cancer treatment by shifting from conventional chemotherapy and radiotherapy toward highly individualized treatment approaches. By genetically engineering a patient’s own T-cells to specifically recognize and attack cancer cells, CAR-T therapy offers higher response rates and more durable clinical outcomes, particularly in patients with relapsed or refractory blood cancers.
The growing effectiveness and adoption of CAR-T therapies are further supported by continuous advancements in genomics, biomarker discovery, and cell engineering technologies. These innovations are enhancing treatment precision, improving patient outcomes, and accelerating the development of next-generation personalized cancer therapies.
Segment Insights
Target Antigen Insights
The CD19-targeted CAR-T Segment Held a 45% Market Share in 2025
The CD19-targeted CAR-T segment dominated the CAR-T therapies for blood cancers market, accounting for a major revenue share of 45% in 2025. This dominance is primarily driven by its strong clinical efficacy and the availability of multiple regulatory approvals for the treatment of leukemia and lymphoma.
CD19 continues to be a highly validated therapeutic target in B-cell malignancies, demonstrating high response rates and durable clinical outcomes, especially in relapsed or refractory cases. Its proven effectiveness and growing adoption across cancer treatment centers worldwide have significantly contributed to the expansion of the segment within the global CAR-T therapies market.
Regional Insights
North America CAR-T Therapies for Blood Cancers Market Size and Growth 2026 to 2035
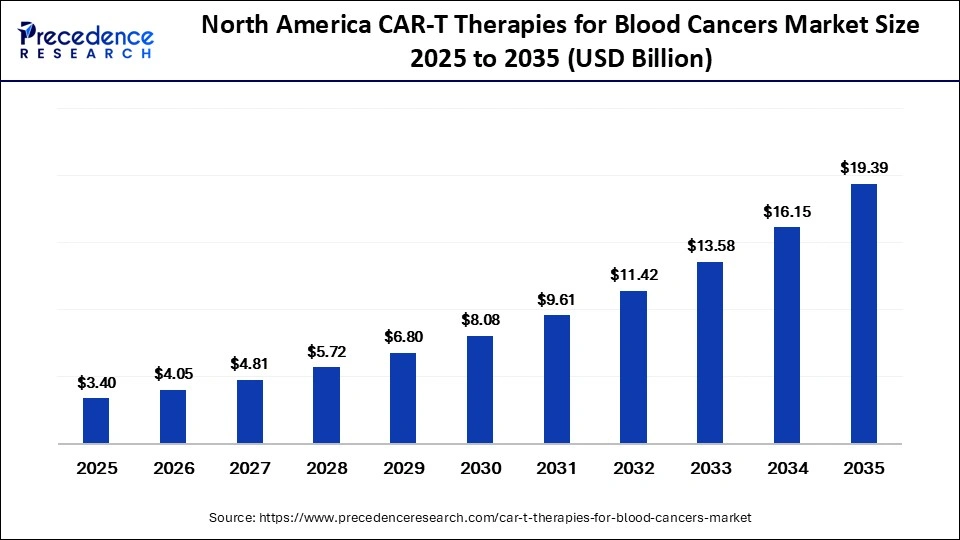
The North America CAR-T therapies for blood cancers market size was valued at USD 3.40 billion in 2025 and is projected to reach approximately USD 19.39 billion by 2035, expanding at a CAGR of 19.02% during the forecast period from 2026 to 2035.
Why Did North America Lead the CAR-T Therapies for Blood Cancers Market in 2025?
North America led the CAR-T therapies for blood cancers market while holding a major share of 50% in 2025, owing to the presence of leading manufacturers of CAR-T therapies. The region also benefits from its well-developed healthcare system and early adoption of innovative therapies. The region accounts for a high burden of blood cancers, including lymphoma and multiple myeloma, supported by an ageing population and improved diagnostic capabilities. Strong clinical research activity, well-established treatment protocols, and a clear regulatory pathway have further facilitated the rapid commercialization and uptake of CAR-T therapies in the region.
U.S. CAR-T Therapies for Blood Cancers Market Size and Growth 2026 to 2035
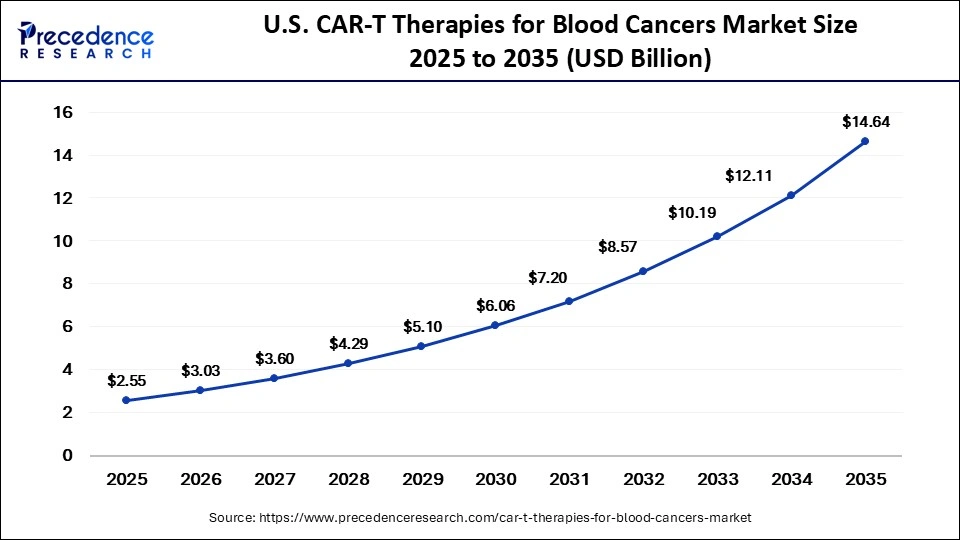
The U.S. CAR-T therapies for blood cancers market size is calculated at USD 2.55 billion in 2025 and is expected to reach nearly USD 14.64 billion in 2035, accelerating at a strong CAGR of 19.10% between 2026 and 2035.
U.S. Market Analysis
The U.S. CAR T-cell therapies for blood cancers market is experiencing significant growth, driven by the rising incidence of hematologic malignancies such as lymphoma and multiple myeloma, alongside an ageing population requiring advanced treatment options. Improvements in diagnostic technologies are enabling earlier detection and timely intervention. A robust healthcare infrastructure, combined with accelerated regulatory approvals and fast-track designations, is supporting wider therapy adoption. Additionally, strong biotech funding and R&D investments are driving continuous innovation in CAR-T development.
CAR-T Therapies for Blood Cancers Market Companies
- Bristol Myers Squibb Company, through its acquisitions of Celgene and Juno Therapeutics, is a leading player in the CAR-T therapies market. The company focuses on developing advanced cell therapies for blood cancers, including innovative treatments for lymphoma and leukemia.
- Johnson & Johnson, in collaboration with Legend Biotech Corporation, is actively advancing CAR-T therapies for multiple myeloma and other hematological malignancies. The partnership has strengthened its position in personalized cancer immunotherapy development.
- Fate Therapeutics, Inc. specializes in developing programmed cellular immunotherapies derived from stem cells. The company is focused on next-generation CAR-T and NK-cell therapies for cancer treatment.
- Allogene Therapeutics, Inc. is dedicated to developing allogeneic CAR-T therapies designed for broader accessibility and faster treatment delivery. Its research emphasizes off-the-shelf cell therapy solutions for blood cancers.
- Cellectis S.A. is known for its expertise in gene-editing technologies and allogeneic CAR-T cell therapies. The company is developing innovative immunotherapies aimed at improving cancer treatment outcomes.
- Bluebird Bio, Inc. focuses on gene and cell therapies for severe genetic diseases and cancers. The company has contributed significantly to advancements in CAR-T and gene-modified cell therapy technologies.
- Autolus Therapeutics plc develops next-generation programmed T-cell therapies for the treatment of cancer. The company emphasizes precision-engineered CAR-T therapies to improve safety, efficacy, and patient outcomes.
Check this report to: Self Driving Bus Market
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Get Sample : https://www.precedenceresearch.com/sample/8377
You can place an order or ask any questions. Please feel free to contact us at sales@precedenceresearch.com |+1 804 441 9344