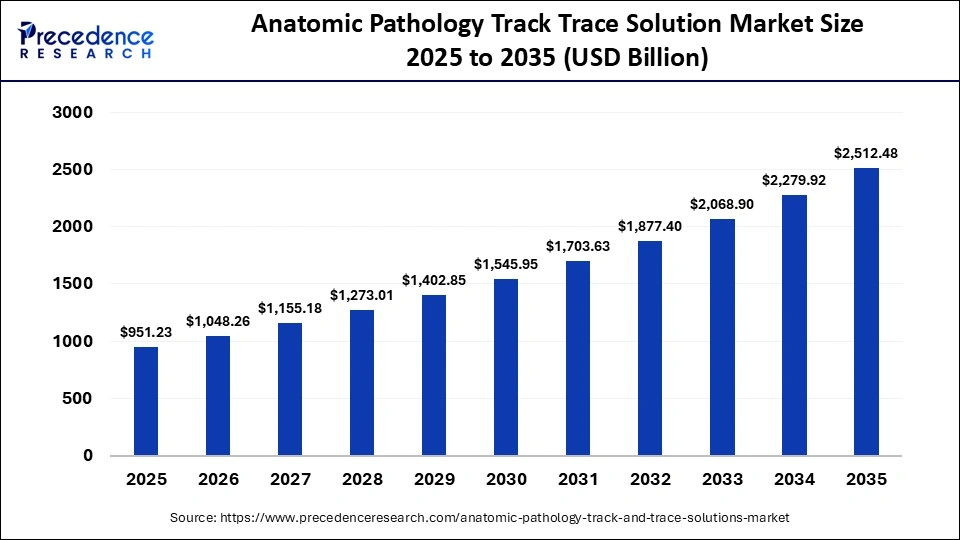
What is the Anatomic Pathology Track Trace Solution Market Size in 2026?
The global anatomic pathology track trace solution market size was accounted for USD 951.23 billion in 2025 and is predicted to increase from USD 1048.26 billion in 2026 to approximately USD 2,512.48 billion by 2035, expanding at a CAGR of 10.20% from 2026 to 2035.Key market drivers include rapid technological innovations, increasing adoption of advanced healthcare technologies, continued digitalization, and rising healthcare awareness.
Key Takeaways
- North America led the anatomic pathology track trace solution market in 2025 with approximately 38.6% share.
- Asia-Pacific is expected to be the fastest-growing region in the forecasted period with about 13.4% CAGR.
- By component, the software segment held a dominant position in the market with a share of 42.7% in 2025.
- By component, the consumables segment is expected to grow at the fastest CAGR of 12.8% in the market between 2026 and 2035.
- By technology, the barcode tracking segment registered its dominance over the global market with a share of 68.4% in 2025.
- By technology, the RFID tracking segment is expected to expand rapidly in the market with a CAGR of 14.1% in the coming years.
- By application, the specimen tracking segment dominated the global market with a share of 36.8% in 2025.
- By application, the chain of custody and compliance tracking segment is expected to grow with the highest CAGR of 13.7% in the market during the studied years.
- By end user, the hospital laboratories segment held the largest revenue share of 47.5% in the market in 2025.
- By end user, the specialty cancer and diagnostic centers segment is expected to witness the fastest growth in the market with a CAGR of 14.4% over the forecast period.
Anatomic Pathology Track & Trace Solution Market Overview
The anatomic pathology track and trace solution market encompasses a wide range of software, hardware, consumables, and integrated workflow systems designed to accurately identify, label, monitor, verify, and document pathology specimens throughout their lifecycle. These solutions support critical processes such as accessioning, grossing, processing, microtomy, staining, archiving, and reporting. By leveraging technologies like barcoding, RFID, and digital workflow automation, these systems enhance specimen traceability, ensure chain of custody, improve patient safety, and reduce errors. They are widely adopted across hospital laboratories, reference labs, academic institutions, and specialized pathology networks to streamline operations, maintain regulatory compliance, and enable real-time visibility and analytics.
Impact of Artificial Intelligence on the Anatomic Pathology Track & Trace Solution Market
Artificial intelligence (AI) is significantly transforming the anatomic pathology track and trace solution market by improving both diagnostic accuracy and operational efficiency. AI-powered image analysis enables pathologists to detect abnormalities with greater precision, while innovations such as virtual staining and synthetic data generation enhance diagnostic capabilities.
In addition, AI automates routine laboratory tasks, including quality control processes and reflex testing, thereby reducing manual workload and minimizing errors. Advanced AI-driven software can extract meaningful insights from high-content imaging data, supporting better decision-making in areas like drug discovery.
Robotic systems further enhance efficiency by handling sample preparation, labeling, and scanning with high accuracy. Moreover, AI algorithms analyze large datasets to predict disease progression and patient response to treatments. Overall, AI plays a crucial role in improving data organization, documentation, and long-term management of pathology workflows, making laboratory operations more intelligent and efficient.
Anatomic Pathology Track Trace Solution Market Trends
- Automated Wax Embedding System: New automated wax embedding modules support standardized histopathology workflow by managing paraffin block preparation efficiently, while enabling operators to focus on accurate tissue orientation in pathology processes.
- Integration of LIMS and Tracking Software: Integration of a laboratory information management system (LIMS) with pathology tracking software improves data security , sample traceability, and workflow efficiency in handling sensitive pathology information.
- Sustainable Track and Trace Solution: Increasing demand for environmentally responsible solutions is encouraging the development of track and trace systems with reduced plastic use, energy efficient operations, and recyclable materials in laboratory settings.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 951.23 Billion |
| Market Size in 2026 | USD 1048.26 Billion |
| Market Size by 2035 | USD 2,512.48 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 10.20% |
| Dominating Region | North America |
| Fastest Growing Region | Asia-Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Component, Technology,Application,End User ,and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Regional Insights
North America holds a leading position in the anatomic pathology track and trace solution market due to its advanced healthcare infrastructure and early adoption of digital pathology systems. Hospitals and diagnostic laboratories in the region are increasingly investing in automation, barcode tracking, and AI-enabled workflow solutions to minimize errors and improve patient safety. Strong regulatory requirements and a focus on compliance further drive the adoption of track and trace systems. Additionally, the presence of major healthcare technology providers and continuous innovation in laboratory automation support market expansion.
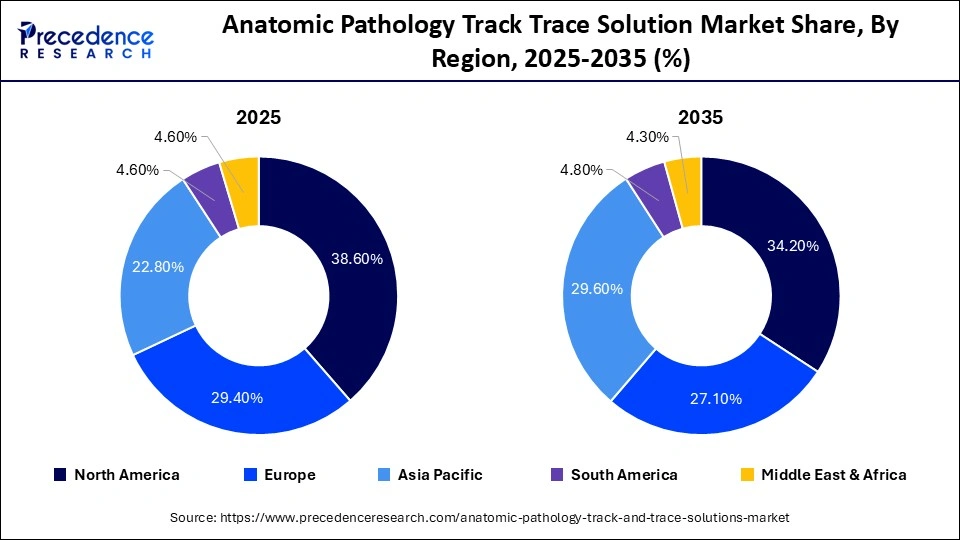
Europe is experiencing steady growth, supported by stringent regulatory frameworks and a strong emphasis on quality assurance in laboratory practices. Countries across the region are actively adopting digital pathology and laboratory information systems to enhance workflow efficiency and traceability. The growing demand for standardized processes and patient safety initiatives is encouraging healthcare providers to implement advanced tracking solutions. Moreover, increasing investments in healthcare digitization and research activities contribute to market growth.
Asia-Pacific is emerging as a rapidly growing market, driven by expanding healthcare infrastructure, rising diagnostic volumes, and increasing awareness of patient safety. Countries such as China, Japan, and India are investing in modern laboratory technologies, including automated pathology systems and digital tracking solutions. The region also benefits from a growing number of diagnostic laboratories and government initiatives to improve healthcare quality, which are accelerating the adoption of track and trace solutions.
Latin America is gradually adopting anatomic pathology track and trace systems, particularly in urban healthcare centers and private laboratories. The market growth is supported by improving healthcare infrastructure and increasing demand for efficient laboratory management systems. While adoption remains at a developing stage, there is growing interest in digital solutions to reduce diagnostic errors and enhance workflow transparency.
The Middle East & Africa region is witnessing gradual growth, driven by investments in healthcare modernization and the development of advanced diagnostic facilities. Countries in the Middle East are focusing on implementing digital health technologies, including pathology tracking systems, to improve service quality. In Africa, adoption is still limited but increasing as healthcare systems continue to evolve and prioritize patient safety and laboratory efficiency.
Anatomic Pathology Track Trace Solution Market Key Players
- Leica Biosystems
- Roche Diagnostics
- Sakura Finetek
- Epredia
- PHC Holdings Corporation
- Epic Systems Corporation
- Hamamatsu Photonics
- Sectra AB
- Indica Labs
- Inspirata
- Gestalt Diagnostics
- Corista
- LigoLab
- NovoPath
- XIFIN
Segments Covered in the Report
By Component
- Software
- Hardware
- Consumables
By Technology
- Barcode Tracking
- RFID Tracking
- Mobile and Cloud Enabled Tracking
By Application
- Specimen Tracking
- Tissue Cassette and Block Tracking
- Slide Tracking
- Chain of Custody and Compliance Tracking
By End User
- Hospital Laboratories
- Independent and Reference Laboratories
- Academic and Research Institutes
- Specialty Cancer and Diagnostic Centers
By Region
- North America
- Latin America
- Europe
- Asia-pacific
- Middle and East Africa
Get Sample Link: https://www.precedenceresearch.com/sample/8247
Also Read : https://www.businesswebwire.com/abdominal-aortic-aneurysm-repair-market/