What is the Pyrogen Testing Market Size in 2026?
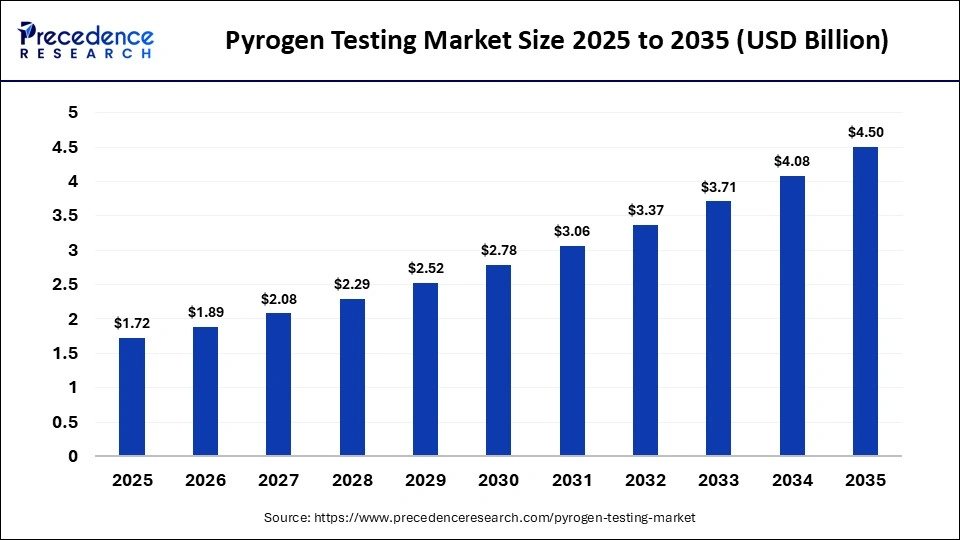
The global pyrogen testing market size was calculated at USD 1.72 billion in 2025 and is predicted to increase from USD 1.89 billion in 2026 to approximately USD 4.50 billion by 2035, expanding at a CAGR of 10.10% from 2026 to 2035. The market is a key indicator of safety for pharmaceutical products, medical devices, and biologics through the identification of fever-inducing contaminants. With regulatory scrutiny being stricter and biologics production growing, pyrogen testing has become the foundation of quality assurance in the life science sector.
Key Takeaways
- North America dominated the global pyrogen testing market, holding a share of 38.60% in 2025.
- Europe is expected to grow at the fastest CAGR in the market during the forecast period.
- By test type, the limulus amoebocyte lysate testing segment held a dominant position in the market by holding a share of 67.80% in 2025.
- By test type, the monocyte activation test segment is expected to grow at the fastest CAGR of 10.80% in the market between 2026 and 2035.
- By offering type, the reagents and kits segment led the global market by holding a share of 51.70% in 2025.
- By offering type, the services segment is expected to grow with the highest CAGR of 9.60% in the market during the studied years.
- By application, the pharmaceutical products testing segment held the largest pyrogen testing market share of 41.60% in 2025.
- By application, the cell and gene therapy products testing segment is expected to expand rapidly in the market with a CAGR of 12.40% in the coming years.
- By end user, the pharmaceutical companies segment registered its dominance over the global market with a share of 46.80% in 2025.
- By end user, the contract research organizations segment is expected to witness the fastest growth in the market with a CAGR of 10.20% over the forecast period.
- By technology, the gel clot LAL method segment contributed the biggest revenue share of 37.20% in the market in 2025.
- By technology, the recombinant factor C assay segment is expected to gain the highest market share with a CAGR of 12.60% between 2026 and 2035.
- By sample type, the injectable drugs segment accounted for the highest revenue share of 38.40% in the market in 2025.
- By sample type, the vaccines segment is expected to show the fastest growth with a CAGR of 11.70% over the forecast period.
Key Factors Driving the Pyrogen Testing Market
The pyrogen testing market is witnessing strong growth due to the increasing complexity of drug formulations and the globalization of pharmaceutical manufacturing. As biologics and advanced therapies become more prevalent, ensuring product safety and sterility has become a top priority for manufacturers.
Modern testing methods such as the Limulus Amebocyte Lysate (LAL) test and recombinant factor C (rFC) assays are gaining widespread adoption due to their high sensitivity and ethical advantages over traditional methods like the Rabbit Pyrogen Test. These advanced solutions not only improve accuracy but also align with regulatory and environmental expectations.
Additionally, leading market players are focusing on innovation, strategic collaborations, and expanding production capacities to meet the growing demand from pharmaceutical and biotechnology industries.
Impact of Artificial Intelligence on Pyrogen Testing
Artificial intelligence (AI) is transforming the pyrogen testing landscape by enhancing precision, efficiency, and predictive capabilities. AI-powered data analytics can identify contamination patterns, optimize testing workflows, and significantly reduce false positives and false negatives in assay results.
Machine learning models are increasingly being used to strengthen quality control by predicting endotoxin contamination risks based on historical datasets. Furthermore, AI-driven automation enables real-time monitoring of manufacturing environments, supporting faster and more informed decision-making.
AI also plays a crucial role in digital validation and regulatory compliance by ensuring consistent documentation, traceability, and streamlined reporting processes key requirements in highly regulated pharmaceutical environments.
Pyrogen Testing Market Trends
- Shift Toward Animal-Free Testing: The market is rapidly transitioning from animal-based methods, such as rabbit pyrogen testing, to in vitro alternatives. Regulatory bodies and ethical considerations are encouraging the adoption of recombinant and synthetic testing solutions.
- Rise of Recombinant Technologies: Recombinant factor C (rFC) assays are emerging as a reliable and sustainable alternative to conventional LAL tests. These methods eliminate dependence on horseshoe crab blood, ensuring supply stability and supporting environmental conservation.
- Automation and Laboratory Digitalization: Increasing automation in pyrogen testing laboratories is reducing human error, improving throughput, and enhancing reproducibility. Advanced instruments and integrated software solutions are streamlining testing workflows and boosting operational efficiency.
- Growth of the Biologics and Vaccine Market: The expanding demand for biologics, vaccines, and cell and gene therapies is significantly increasing the need for highly sensitive pyrogen detection methods due to the complexity of these products.
- Rising Outsourcing to CROs: Pharmaceutical companies are increasingly outsourcing pyrogen testing to contract research organizations (CROs) to reduce costs, access specialized expertise, and accelerate time-to-market.
- Increasing R&D Investments: Growing investments in drug discovery and development are driving demand for advanced quality control solutions, including pyrogen testing, across all stages of the pharmaceutical pipeline.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 1.72 Billion |
| Market Size in 2026 | USD 1.89 Billion |
| Market Size by 2035 | USD 4.50 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 10.10% |
| Dominating Region | North America |
| Fastest Growing Region | Europe |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Test Type, Offering Type, Application, End User, Technology, Sample Type, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Pyrogen Testing Market Regional Insights
North America dominates the global pyrogen testing market, accounting for the largest revenue share (over 30%+). This leadership is driven by a strong presence of pharmaceutical and biotechnology companies, advanced healthcare infrastructure, and strict regulatory frameworks such as those enforced by the FDA. The region also benefits from high adoption of advanced in vitro testing methods, increasing biologics production, and continuous R&D investments, making it a key hub for innovation in pyrogen testing.
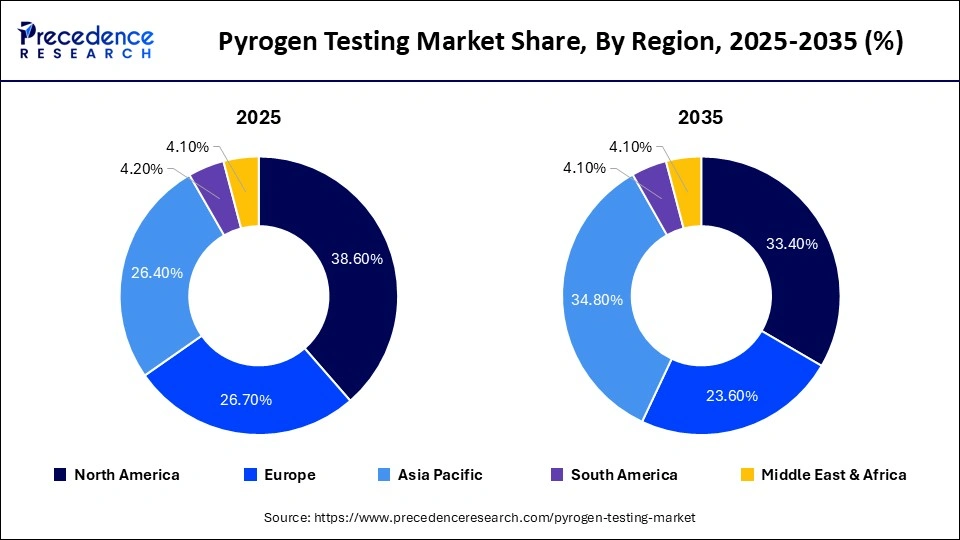
Europe holds a significant share in the pyrogen testing market due to its well-established pharmaceutical industry and stringent regulatory environment governed by the European Medicines Agency (EMA). The region is witnessing increased adoption of ethical, animal-free testing methods such as recombinant factor C (rFC) and Monocyte Activation Tests (MAT). Growing investments in drug development and strong emphasis on patient safety are further accelerating market growth.
Asia Pacific is the fastest-growing region in the pyrogen testing market, with a projected high CAGR during the forecast period. Growth is fueled by expanding pharmaceutical manufacturing in countries such as China, India, and Japan, rising healthcare expenditure, and increasing regulatory enforcement for drug safety. The region is also emerging as a global hub for clinical trials and biomanufacturing, significantly boosting demand for advanced pyrogen testing solutions.
Pyrogen Testing Market Value Chain Analysis

Pyrogen Testing Market Key Players
- Charles River Laboratories
- Lonza Group
- Thermo Fisher Scientific
- Merck KGaA
- BioMérieux
- Associates of Cape Cod
- Wako Chemicals USA
- GenScript
- Creative Bioarray
- Hyglos GmbH
- Microcoat Biotechnologie
- Ellab A/S
- Seikagaku Corporation
- Sanquin Reagents
- Fujifilm Wako Chemicals
Recent Developments
- In February 2026, the EDQM and EPAA hosted the Pyrogen Testing 2.0 symposium, focusing on ethical, rapid, and sustainable non-animal pyrogen testing methods. The event was hosted to promote state-of-the-art approaches for safe pyrogen detection globally, reflecting evolving practices in the industry.
- In January 2026, Betmat launched high-performance, sustainable endotoxin testing services to address the evolving needs of the industry. This initiative focuses on delivering reliable and efficient testing solutions while emphasizing environmental sustainability, positioning Betmat as a leader in quality assurance for pharmaceuticals and biologics.
Segment Covered in the Report
By Test Type
- Limulus Amebocyte Lysate testing
- Monocyte Activation Test
- Rabbit Pyrogen Test
By Offering Type
- Reagents and Kits
- Instruments and Readers
- Consumables
- Services
By Application
- Pharmaceutical Products Testing
- Biologics and Biotechnology Products Testing
- Medical Devices Testing
- Cell and Gene Therapy Products Testing
- Vaccine Testing
By End User
- Pharmaceutical Companies
- Biotechnology Companies
- Contract Research Organizations
- Academic and Research Institutes
- Medical Device Manufacturers
By Technology
- Gel Clot LAL Method
- Turbidimetric LAL Method
- Chromogenic LAL Method
- Recombinant Factor C Assay
By Sample Type
- Injectable Drugs
- Implantable Medical Devices
- Parenteral Nutrition Products
- Dialysis Products
- Vaccines
By Region
- North America
- Latin America
- Europe
- Asia-pacific
- Middle and East Africa
Get Free Sample Link: https://www.precedenceresearch.com/sample/8200