What is the Catheter Stabilization Device Market Size in 2026?
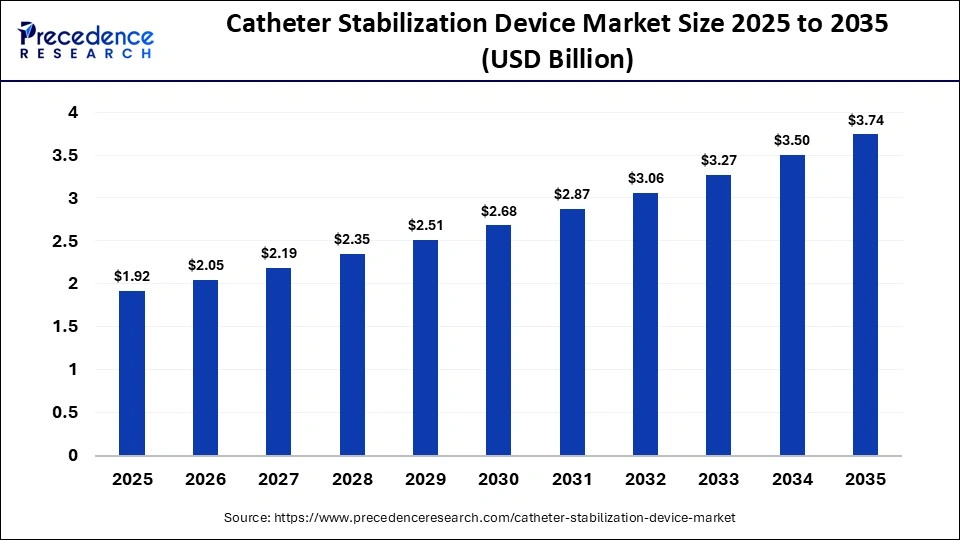
The global catheter stabilization device market size was calculated at USD 1.92 billion in 2025 and is predicted to increase from USD 2.05 billion in 2026 to approximately USD 3.74 billion by 2035, expanding at a CAGR of 6.90% from 2026 to 2035. The market is driven by the rising need to prevent catheter-related complications and infections, growing hospitalizations, and advancements in patient-centric securement technologies.
Key Takeaways
- North America dominated the global catheter stabilization device market with a share of 37.80% in 2025.
- The Asia Pacific is expected to grow at the fastest CAGR in the market during the forecast period.
- By product type, the peripheral intravenous catheter stabilization devices segment held a dominant position in the market with a share of 31.80% in 2025.
- By product type, the central venous catheter stabilization devices segment is expected to grow at the fastest CAGR of 8.0% between 2026 and 2035.
- By mechanism type, the adhesive securement devices segment held a 34.70% share of the market in 2025.
- By mechanism type, the locking securement devices segment is expected to grow with the highest CAGR of 9.20% during the studied period.
- By material, the polyurethane-based devices segment held a dominant position in the market, accounting for 37% share of the market in 2025.
- By material, the silicone-based devices segment is expected to grow at the fastest CAGR of 8.50% from 2026 to 2035.
- By end user, the hospitals segment led the global market with the largest share of 68.60% in 2025.
- By end user, the home care settings segment is expected to expand rapidly in the market with a CAGR of 9.50% in the coming years.
Catheter Stabilization Device Market Overview
The catheter stabilization device market includes a wide range of medical securement solutions designed to anchor catheters, tubes, and drainage lines to the skin. These devices help prevent catheter movement, accidental dislodgement, and complications such as infections, ensuring improved patient safety and clinical outcomes.
The market comprises adhesive-based, sutureless, locking, strap-based, and anchor-pad systems used across various applications, including peripheral, central venous, arterial, urinary, epidural, and chest drainage catheters. These devices are widely utilized in hospitals, ambulatory care centers, and home healthcare settings.
Market growth is primarily driven by the rising prevalence of chronic diseases such as cardiovascular disorders, cancer, and kidney diseases, which increase the need for catheterization procedures. Additionally, the growing emphasis on patient comfort and safety, along with advancements in skin-friendly adhesives, antimicrobial materials, and sutureless fixation technologies, is further fueling market expansion.
Impact of Artificial Intelligence on the Catheter Stabilization Device Market
Artificial intelligence (AI) is playing an increasingly important role in transforming the catheter stabilization device market by enhancing patient care and enabling smart monitoring capabilities.
AI-powered systems can continuously track catheter positioning and detect early signs of dislodgement or complications. This allows healthcare providers to receive real-time alerts and take prompt preventive action, reducing the risk of adverse events.
Furthermore, AI is being integrated into the development of next-generation stabilization devices with improved adhesion, durability, and skin compatibility. These innovations enhance patient comfort while minimizing device-related complications.
As healthcare systems move toward connected and data-driven environments, AI is helping streamline clinical workflows, improve decision-making, and drive innovation in catheter stabilization technologies.
Catheter Stabilization Device Market Trends
- Rising Adoption of Sutureless Devices: Healthcare professionals are increasingly adopting sutureless catheter stabilization devices to reduce needlestick injuries, enhance patient comfort, and minimize infection risks. These devices align with strict safety protocols and regulatory standards across healthcare settings.
- Advancements in Antimicrobial and Skin-Friendly Materials: Manufacturers are focusing on developing advanced materials such as antimicrobial coatings and hypoallergenic adhesives. These innovations help prevent infections and reduce skin irritation, particularly for patients requiring long-term catheterization.
- Growth of Home Healthcare and Ambulatory Care: The expansion of home healthcare and ambulatory services is driving demand for easy-to-use and reliable catheter stabilization solutions. These devices support safe long-term catheter management outside hospital settings, helping reduce healthcare costs and hospital readmissions.
- Focus on Reducing Catheter-Related Complications: With stricter clinical guidelines in place, there is increasing demand for advanced securement systems that minimize catheter movement and reduce the risk of catheter-associated bloodstream infections (CABSI). This trend reflects the broader focus on improving patient outcomes through medical device innovation.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 1.92 Billion |
| Market Size in 2026 | USD 2.05 Billion |
| Market Size by 2035 | USD 3.74 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 6.90% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Product Type, Mechanism Type, Material, End User, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Regional Insights
North America dominates the catheter stabilization device market, accounting for the largest share of approximately 33-38% globally. This leadership is driven by advanced healthcare infrastructure, high surgical volumes, and widespread adoption of catheterization procedures. Strong regulatory standards focused on infection prevention and patient safety, along with high healthcare spending, further support market growth. Additionally, the increasing prevalence of chronic diseases and the presence of leading medical device manufacturers contribute to the region’s dominance.
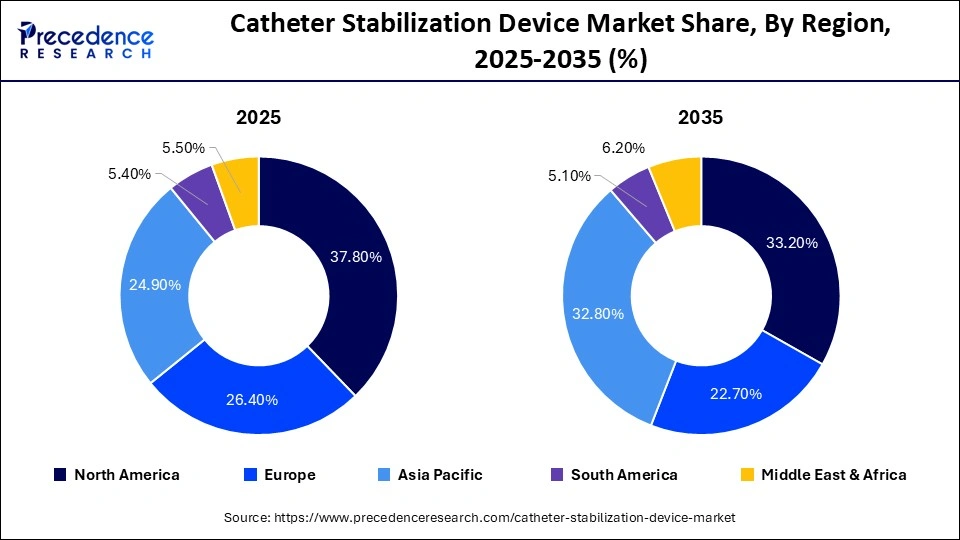
Europe holds a significant share of around 22-26% of the global market. The region benefits from well-established healthcare systems, stringent regulatory frameworks, and a strong emphasis on infection control practices. Countries such as Germany, the UK, and France are key contributors, with widespread adoption of advanced catheter securement technologies. Rising aging populations and increasing surgical procedures further drive demand.
Asia Pacific is the fastest-growing region in the catheter stabilization device market, accounting for over 24-32% of the global share. Rapid growth is fueled by expanding healthcare infrastructure, rising population, and increasing prevalence of chronic diseases. Countries such as China, India, and Japan are witnessing strong demand due to growing hospital networks, government healthcare initiatives, and increasing awareness of infection prevention. The region is also benefiting from the rise of medical tourism and contract manufacturing.
The Middle East & Africa region is gradually emerging, contributing global market. Growth in this region is driven by expanding healthcare infrastructure, increasing government initiatives, and rising demand for advanced medical devices. Countries such as the UAE, Saudi Arabia, and South Africa are leading adoption, although challenges such as limited access in rural areas remain.
Catheter Stabilization Devices Market Value Chain Analysis
- Raw Material Sourcing
This stage involves sourcing essential raw materials such as medical-grade adhesives, polymers, silicone, and nonwoven fabrics, which are critical for ensuring patient safety, comfort, and durability.
Key Players:Dow Inc., DuPont, and 3M
- Component Manufacturing
At this stage, raw materials are processed into components such as adhesive backings, securement bases, locking mechanisms, and skin-friendly interfaces.
Key Players: Avery Dennison Corporation, Scapa Group plc, and Berry Global Inc.
- Clinical Trial and Regulatory Authorizations
Innovations are highly regulated under clinical trials to establish the safety and performance, along with the compatibility of the devices with various kinds of catheters. Regulatory approvals like the FDA and EMA given to the product before its market introduction are valid guarantees that the product conforms to the international standards of quality, safety, and efficacy.
Key Players: B. Braun Medical Inc., Smiths Medical, 3M Company
- Distribution to Hospitals and Pharmacies
Finished equipment is distributed via hospital procurement, medical distributors, and pharmacies. Efficient logistics will ensure timely delivery, maintenance of product integrity, and ease of delivery of the products in the inpatient, outpatient, and home health units.
Key Players: McKesson Corporation, Cardinal Health, Inc., Medline Industries, Inc.
Catheter Stabilization Device Market Companies
- Becton, Dickinson and Company
- 3M
- Baxter International
- B. Braun Melsungen AG
- Cardinal Health
- ConvaTec
- Merit Medical Systems
- Medtronic
- ICU Medical
- Dale Medical Products
- Centurion Medical Products
- TIDI Products
- Teleflex Incorporated
- Vygon
- Avanos Medical
Recent Developments
- In January 2025, the B. Braun medical company came up with a low-profile, soft catheter under the brand name of Clik-FIX Epidural/Peripheral Nerve Block Catheter Securement Device, which is designed to reduce catheter motion and dislocation in case of regional anesthesia. This innovation is expected to enhance patient safety and pain management during operations.
- In February 2024, Cook Medical merged with Bedal International to purchase FlexGRIP catheter securement devices as an addition to its percutaneous drainage lines. The collaboration will promote patient care and mobility throughout various treatment procedures
Segments Covered in the Report
By Product Type
- Peripheral intravenous catheter stabilization devices
- Central venous catheter stabilization devices
- Arterial catheter stabilization devices
- Urinary catheter stabilization devices
- Chest drainage tube stabilization devices
- Epidural catheter stabilization devices
- All-site catheter stabilization devices
By Mechanism Type
- Adhesive securement devices
- Sutureless securement devices
- Anchor pad securement devices
- Locking securement devices
- Strap-based securement devices
By Material
- Silicone-based devices
- Foam-based devices
- Fabric-based devices
- Hydrocolloid-based devices
- Polyurethane-based devices
By End User
- Hospitals
- Ambulatory surgical centers
- Home care settings
- Specialty clinics
By Region
- North America
- Latin America
- Europe
- Asia-pacific
- Middle and East Africa
Get Sample Link: https://www.precedenceresearch.com/sample/8195